Radium bromide illuminated by its own radioactive glow. Marie Curie, in turn, died of what was almost certainly radiation poisoning. Pierre Curie carried a vial of radium in his coat pocket to demonstrate its greenish glow, a habit that caused him to become ill from radiation poisoning well before he was run over by a horse-drawn wagon and killed instantly in 1906. 
Starting with several tons of pitchblende, the Curies isolated two new radioactive elements after months of work: polonium, which was named for Marie’s native Poland, and radium, which was named for its intense radioactivity. She found that one particular uranium ore, pitchblende, was substantially more radioactive than most, which suggested that it contained one or more highly radioactive impurities. (from the Latin radius, meaning “ray”) to describe the emission of energy rays by matter. Marie Curie coined the term radioactivity The spontaneous emission of energy rays (radiation) by matter. Becquerel’s work was greatly extended by Marie Curie (1867–1934) and her husband, Pierre (1854–1906) all three shared the Nobel Prize in Physics in 1903. The second line of investigation began in 1896, when the French physicist Henri Becquerel (1852–1908) discovered that certain minerals, such as uranium salts, emitted a new form of energy. Most important for chemistry, Thomson found that the mass-to-charge ratio of cathode rays was independent of the nature of the metal electrodes or the gas, which suggested that electrons were fundamental components of all atoms. Because like charges repel each other and opposite charges attract, Thomson concluded that the particles had a net negative charge we now call these particles electrons. These particles were emitted by the negatively charged cathode and repelled by the negative terminal of an electric field. More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles. He demonstrated that cathode rays could be deflected, or bent, by magnetic or electric fields, which indicated that cathode rays consist of charged particles ( Figure 1.17 "Deflection of Cathode Rays by an Electric Field"). Thomson (1856–1940) proved that atoms were not the ultimate form of matter. Researchers trying to understand this phenomenon found that an unusual form of energy was also emitted from the cathode, or negatively charged electrode hence this form of energy was called cathode rays. Long before the end of the 19th century, it was well known that applying a high voltage to a gas contained at low pressure in a sealed tube (called a gas discharge tube) caused electricity to flow through the gas, which then emitted light ( Figure 1.16 "A Gas Discharge Tube Producing Cathode Rays"). When a high voltage is applied to a gas contained at low pressure in a gas discharge tube, electricity flows through the gas, and energy is emitted in the form of light. 
In fact, the elucidation of the atom’s structure is one of the greatest detective stories in the history of science.įigure 1.16 A Gas Discharge Tube Producing Cathode Rays 
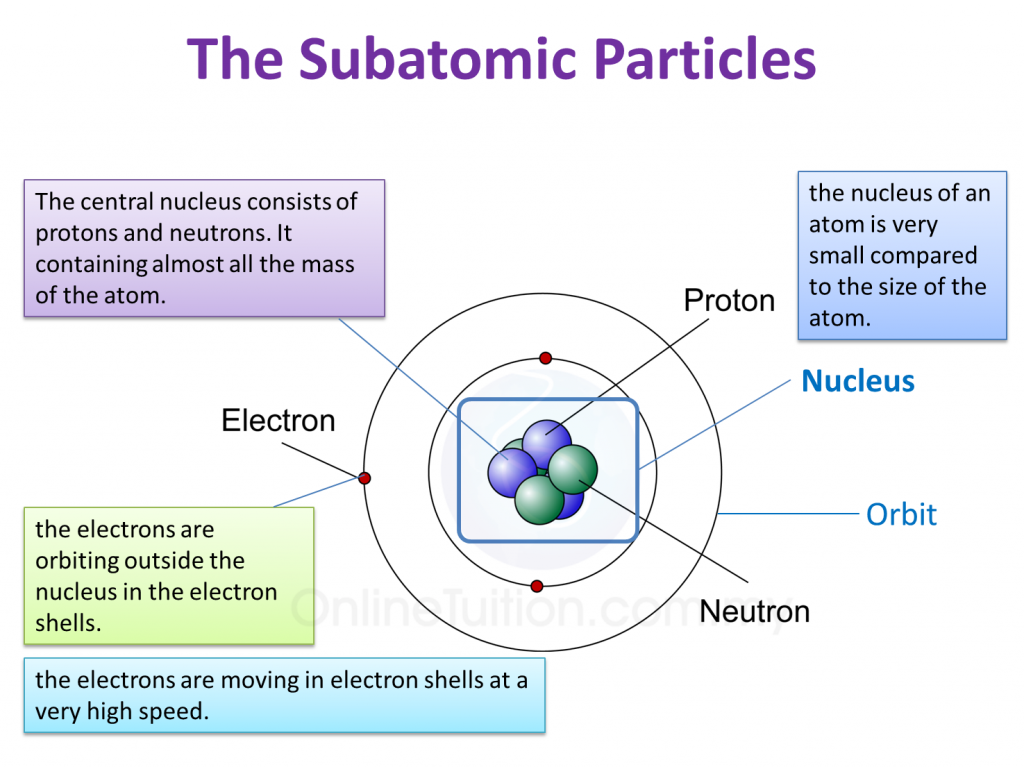
The discovery of the electron and the proton was crucial to the development of the modern model of the atom and provides an excellent case study in the application of the scientific method. Protons and neutrons constitute by far the bulk of the mass of atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
